Description
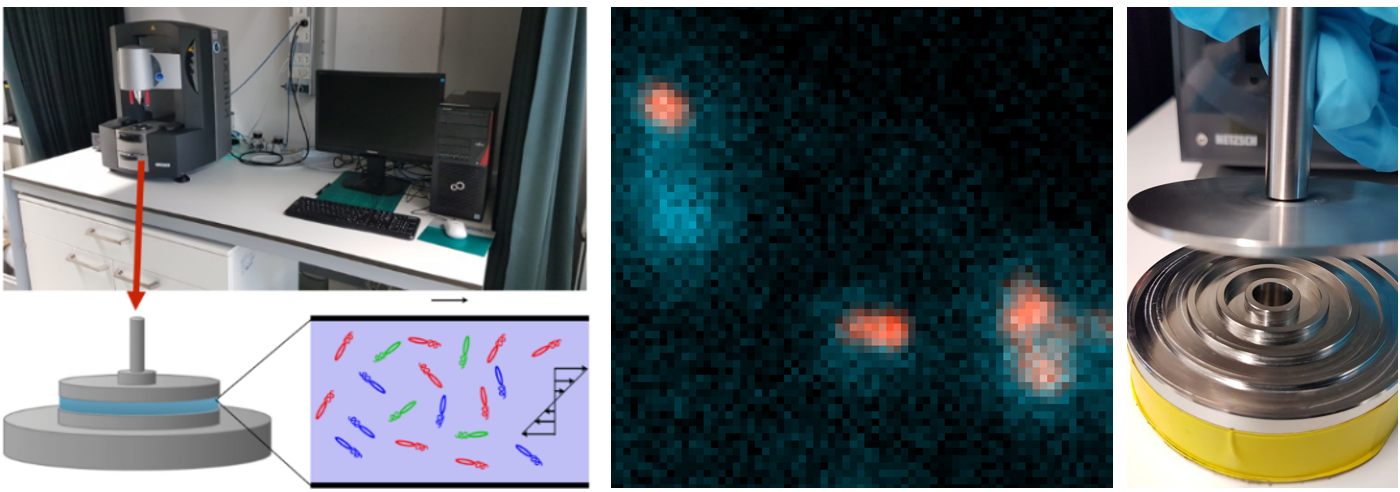
Bacteria evolve rapidly because they can share genes not only vertically, from parents to offspring during division, but also horizontally, directly between each other.This ability to exchange genes contributes to the spread of antibiotic resistance in the environment, a major emerging public health hazard that could cause 10 million deaths each year by 2050. Conjugation, a key mechanism of environmental gene transfer,involves two steps (see figure). First, an encounter between bacteria needs to occur to establish direct cell–cell contact. Second, transfer of genetic material, such as a resistance-carrying plasmid, takes place through a pilus, a ‘syringe’ through which the material is injected from the donor cell to the recipient cell. Crucially, both steps need to be quantified to accurately characterize conjugation and thus the spread of genetic information. However, to date, these two steps have been treated as one. As a result, it remains unknown whether high or low gene transfer rates originate from frequent encounters or an efficient conjugation mechanism, making it challenging to predict the rates with which genes spread in microbial ecosystems.
In this thesis, the student will carry out experiments to measure the key missing parameter that characterizes gene exchange between bacteria, the probability that a bacterium-bacterium encounter results in a gene (plasmid) exchange. The student will carefully mix two bacterial strains in a fluid using a rheometer to control cell-cell encounters, and use fluorescence microscopy backed up with machine learning to count how many gene transfer events occurred (see figure). During the project, the student will learn how to culture bacteria, perform conjugation experiments and investigate the impact of fluid mixing on the rates of gene exchange between bacteria.
Goal
Completion of the development of a method to measure the frequency of bacterial conjugation events as a function of the conjugation rate and its application in varying conditions.
Skills you will learn
• Culture and grow bacteria
• Handling of a rheometer
• Performing of conjugation experiments
• Fluorescence microscopy
• Microscopy image analysis
• Critical thinking and protocol optimization
Project start
September 2024
Location
Environmental Microfluidics Laboratory, IfU, D-BAUG (ETH Zurich, Hoenggerberg campus)
Project type
This project can be adapted for Master Thesis or Master Project.
Contact details
The thesis will be supervised by Dr. Jonasz Słomka and Prof. Roman Stocker and will be carried out at the Institute of Environmental Engineering.
For more information, please contact Dr. Jonasz Słomka (slomka@ifu.baug.ethz.ch).